Which of the following is the correct observation of the reaction shown in the following set up?
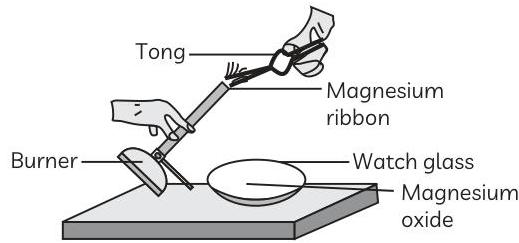
(a) Brown powder of Magnesium oxide is formed.
(b) Colourless gas which turns lime water milky is evolved.
(c) Magnesium ribbon burns with brilliant white light.
(d) Reddish brown gas with a smell of burning Sulphur has evolved.
[CBSE SQP 2022]
Ans. (c) Magnesium ribbon burns with brilliant white light.
Explanation: Magnesium ribbon burns with a dazzling white light and produces a white powder, which is magnesium oxide as shown in the figure. The reaction can be represented as:
(White powder)