Read the following passages and answer the questions that follow:
Rahul was asked to perform an experiment by his science teacher. The teacher told Rahul to take some copper powder in a china dish and heat it by placing it on the Bunsen burner with the help of a wire gauze and tripod stand. After some time, Rahul observed that the copper powder had changed its colour.
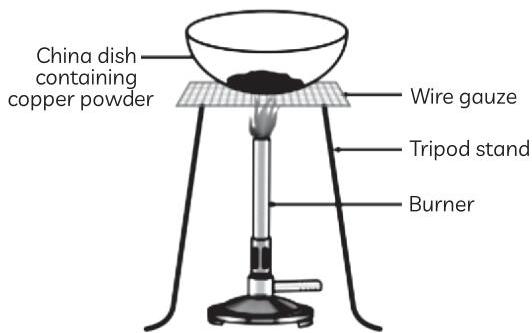
(A) Observe the given figure and write what happened when Rahul heated the copper powder in a china dish. Explain his observation.
(B) What will you observe if hydrogen gas is passed over the heated material obtained in (A) above?
(C) (i) Identify the substance oxidised and substance reduced in the reaction at (B) above.
(ii) Given is the reaction between lead sulphide and hydrogen peroxide shown by the equation below.
Identify oxidised and reduced compounds.
Ans. (A) When copper powder is heated in a china dish by Rahul, the surface of copper powder is coated with black copper oxide as oxidation of copper takes place by addition of oxygen to copper.
(B) When hydrogen gas is passed over heated CuO, the black coating on the surface turns brown, as reverse reaction takes place and copper is formed.
(C) (i) In the reaction (B), hydrogen is oxidised to water by addition of oxygen, whereas copper oxide is reduced to copper by loss of oxygen.
(ii) Lead sulphide is oxidised and hydrogen peroxide is reduced.
In the reaction given, lead sulphide gains oxygen and therefore oxidises to form lead sulphate, whereas hydrogen peroxide loses oxygen and reduces to form water.