The emission of brown fumes in the given experimental set-up is due to:
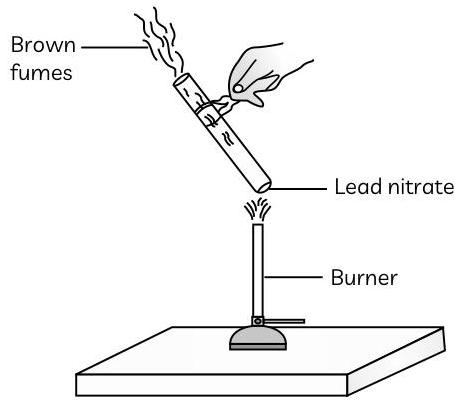
(a) thermal decomposition of lead nitrate which produces brown fumes of nitrogen dioxide.
(b) thermal decomposition of lead nitrate which produces brown fumes of lead oxide.
(c) oxidation of lead nitrate forming lead oxide and nitrogen dioxide.
(d) oxidation of lead nitrate forming lead oxide and oxygen. [CBSE 2023]
Ans. (a) thermal decomposition of lead nitrate which produces brown fumes of nitrogen dioxide.
Explanation: We see brown vapours in the given experiment when we heat lead nitrate powder in a boiling test tube. Lead nitrate undergoes a decomposition reaction when heated, resulting in the formation of lead oxide, nitrogen dioxide, and oxygen. The reaction is:
The brown coloured fumes are of nitrogen dioxide.