Question
Study the figure given below and answer the following questions:
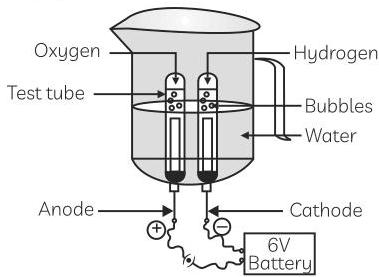
(A) Name the process depicted in the diagram.
(B) Write the composition of gases collected at anode and cathode.
(C) Write the balanced chemical equation of the reaction taking place in this case.
(D) The reaction does not take place if a few drops of dilute sulphuric acid are not added to water. Why? [CBSE 2020]
Verified Answer
Ans. (A) Electrolytic decomposition of water/electrolysis of water.
(B) The gas collected at cathode is hydrogen which is double the volume of oxygen collected at anode.
Helpful?