## Example 2. Case Based:
A 2 cm long thin ribbon of a metal 'X' was taken and first cleaned with a sandpaper. It was then burnt using a spirit lamp or burner by holding it with a pair of tongs. The ribbon burnt with a dazzling white flame and formed a powder 'Y' which was collected in a watch glass.
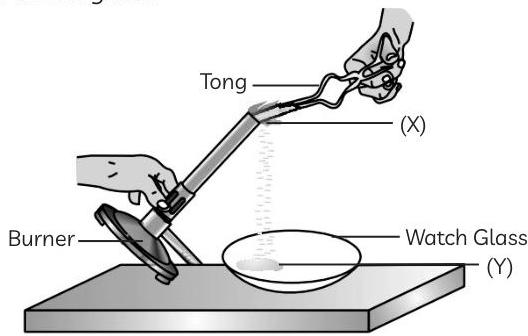
(A) Which option correctly identifies both (X) and (Y)?
(Analyse)
(B) The colour of powder or ash formed when a magnesium ribbon is burnt in air is:
(a) grey
(b) black
(c) white
(d) yellow (Remember)
(C) Why should a magnesium ribbon be cleaned before burning in air? (Understand)
[NCERT]
(D) Why is it advisable to wear eye protection while burning magnesium ribbon?
(Understand)
(E) Assertion (A): Magnesium ribbon burns with a dazzling white flame.
Reason (R): When magnesium ribbon burns in air, only heat is evolved.
(a) Both (A) and (R) are true, and (R) is the correct explanation of (A).
(b) Both (A) and (R) are true, and (R) is not the correct explanation of (A).
(c) (A) is true but (R) is false.
(d) (A) is false but (R) is true. (Remember)
Ans. (A) (c) (X): Magnesium; (Y): Magnesium oxide
Explanation: When magnesium ribbon is burnt, it forms a white powder of magnesium oxide (MgO) due to the reaction between magnesium and oxygen present in the air.
(B) (c) white
Explanation: When a magnesium ribbon is burnt in air, it forms magnesium oxide, which is a white coloured powder.
(C) Magnesium ribbon should be cleaned before burning in air to remove the layer of magnesium oxide that may have formed on the ribbon due to the reaction of magnesium with oxygen. This white layer of magnesium oxide prevents the magnesium ribbon from burning.
(D) When magnesium ribbon is burned, a brilliant white light is formed. This light contains ultraviolet (UV) radiation, which can cause permanent eye damage. Wearing eye protection is therefore advised when burning the magnesium ribbon.
(E) (c) (A) is True but (R) is False.
Explanation: Magnesium ribbon burns in the presence of air to form magnesium oxide (). A large amount of heat and light (containing UV radiation) are produced, resulting in a dazzling white flame.