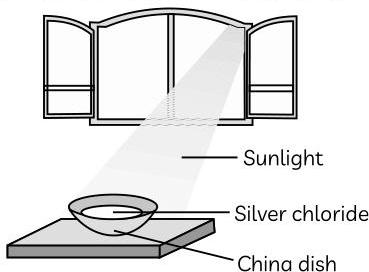
Aditya was tasked with the clean up and organisation of the chemistry lab by his teacher. However, he forgot to put the silver chloride back into the black plastic bottle, and as a result of this, the silver chloride was left in a china dish near the window of the laboratory, where sunlight is falling directly upon it. When Aditya arrived the next day, he observed that the colour of silver chloride had changed and that it was now looking like a greyish powder. His teacher told him that silver containing compounds are usually stored in black plastic bottles so that they can be prevented from undergoing decomposition. He also told Aditya that chlorine was released during this reaction.
(A) Why the colour change was observed in the china dish by Aditya?
(B) What name is given to such type of reactions?
(C) On heating marble, a product 'P' is formed whose solution is used for white-washing the walls. Identify 'P'. Write the equations for:
(A) Why the colour change was observed in the china dish by Aditya?
(B) What name is given to such type of reactions?
(C) On heating marble, a product 'P' is formed whose solution is used for white-washing the walls. Identify 'P'. Write the equations for:
(A) Why the colour change was observed in the china dish by Aditya?
(B) What name is given to such type of reactions?
(C) On heating marble, a product 'P' is formed whose solution is used for white-washing the walls. Identify 'P'. Write the equations for:
(i) heating of marble.
(ii) formation of solution of 'P'.
Ans. (A) This sunlight causes the decomposition of silver chloride (which is white in colour) into grey silver and chloride gas is given off.
(B) This type of reaction is photochemical decomposition reaction. Chemical reactions carried out in the presence of sunlight are also known as photolytic decomposition reactions.
(C) 'P' is calcium oxide (CaO).
(i) \n\n(ii)