A synthesis or combination reaction is a reaction in which simple compounds combine to make a more complex one. The opposite of combination reaction is a decomposition reaction in which a single substance splits into two or more substances. We also have
single displacement reaction (in which a more reactive element displaces a less reactive element from its compound) and double displacement reaction (in which exchange of ions takes place between two compounds). Having a thorough understanding of the types of reactions will be useful for predicting the products of an unknown reaction.
(A) Prashant performed several experiments and then noted down his observations in a tabular form as given:
| S. No. | Observation | Conclusions Regarding types of Reactions Taking Place |
|---|---|---|
| (I) | When copper powder is heated in a china dish, the surface of copper powder turns black. | Combination reaction |
| (II) | Hydrogen gas is evolved when iron reacts with dil. HCl. | Decomposition reaction |
| (III) | A black precipitate is formed along with sulphuric acid solution when hydrogen sulphide gas is passed through copper sulphate solution. | Double displacement reaction |
| (IV) | When electricity is passed through molten aluminium chloride, aluminium metal is formed along with a gas. | Displacement reaction |
Select the option that correctly describes the conclusions made by the student regarding type of reaction taking place:
(a) Both (I) and (II)
(b) Both (I) and (III)
(c) Both (II) and (III)
(d) (I), (III) and (IV)
(B) When a metal X, which is used for coating iron articles, is added to a blue solution of a reddish brown metal Y, the colour of the solution gets discharged.
Select the option which correctly identifies the metals X and Y and the type of reaction taking place.
| Option | Metal X | Metal Y | Type of Reaction |
|---|---|---|---|
| (a) | Zinc | Iron | Double displacement |
| (b) | Copper | Zinc | Double displacement |
| (c) | Copper | Zinc | Displacement |
| (d) | Zinc | Copper | Displacement |
(C) Assertion (A): The reaction between copper oxide and hydrogen is an oxidation reduction reaction.
Reason (R): Copper oxide acts as an oxidising agent and hydrogen acts as a reducing agent.
(a) Both (A) and (R) are true, and (R) is the correct explanation of (A).
(b) Both (A) and (R) are true, and (R) is not the correct explanation of (A).
(c) (A) is true but (R) is false.
(d) (A) is false but (R) is true.
(D) Which of the following statement(s) are INCORRECT?
(I) The precipitation reactions produce insoluble salts.
(II) Displacement reactions are opposite of combination reactions.
(III) Decomposition reactions can be carried out only in presence of heat or light.
Options:
(a) Only (I)
(b) Only (III)
(c) Both (I) and (III)
(d) Both (II) and (III)
OR
(D) Ravi placed copper sulphate solution in an iron vessel and Mahima kept iron sulphate solution in a copper vessel for a few days. In which vessel the holes can be seen?
(a) Iron vessel
(b) Copper vessel
(c) Both (a) and (b)
(d) Holes will not appear in any vessel
Ans. (C) (a) Both (A) and (R) are true, and (R) is the correct explanation of (A).
Explanation: The reaction between copper oxide and hydrogen to form copper and water is an oxidation-reduction reaction which is also a displacement reaction.
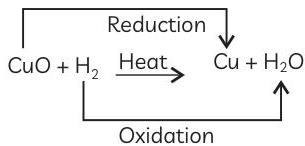
In this reaction, CuO is changing to Cu so copper oxide is being reduced to copper. H₂ is changing into water (H₂O) so hydrogen is being oxidised to water. CuO is giving the oxygen required for the oxidation of hydrogen, therefore, copper oxide is the oxidising agent. Hydrogen is responsible for removing oxygen from copper oxide, therefore, hydrogen is the reducing agent.
(D) (d) Both (II) and (III)
Explanation: Decomposition reactions require energy in the form of heat, light or electricity. Decomposition reactions are the opposite of combination reactions.
(D) (a) Iron vessel
Explanation: Ravi would see holes in an iron vessel as iron is more reactive than copper and hence, displaces copper from its (CuSO₄) solution.