53. During the time period of her experimental class, the science teacher Mrs. Priyanjali, placed an experimental set-up in front of the students as shown in figure (a).
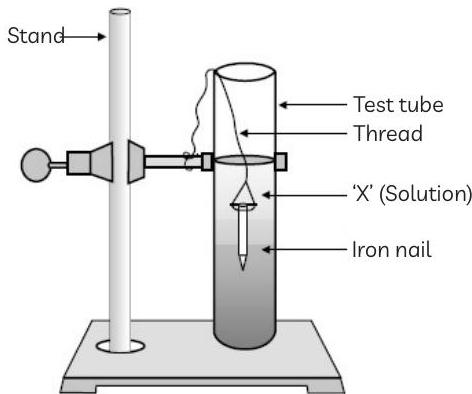

It was observed that iron nails are immersed in a solution of a compound 'P'. After a few hours, the students observed the change in the iron nail as shown in the figure (b).
(A) Identify the chemical compound 'P'. What change did the students observe in the colour of solution of compound 'P'? Why did this change occur?
(B) Shreya, one of the students from class was trying to figure out the type of reaction that took place in the given setup. Help her by identifying the same and give reason why is this reaction classified as such?
(C) After the experiment, the teacher told Shreya that she will demonstrate the same experiment next time with nails of two different metals. Will the same reaction take place if:
(i) Zinc nails are immersed in copper sulphate?
(ii) Copper nails are immersed in copper sulphate solution?
Ans. (A) Compound 'P' is copper sulphate or . The colour of the copper sulphate solution changes from blue to light green, because iron being more reactive than copper displaces copper from copper sulphate solution and forms ferrous sulphate solution (green). Copper gets deposited on iron nail.
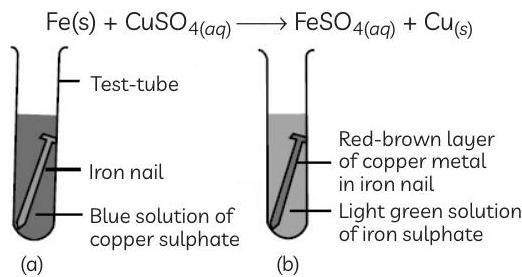
(B) It is a single displacement reaction because iron displaces copper from copper sulphate solution.
(C) (i) Zinc nails will be able to displace copper from its respective solution and thus, the reaction will take place.
(ii) No reaction will take place in this case, as copper cannot displace itself from its own solution.